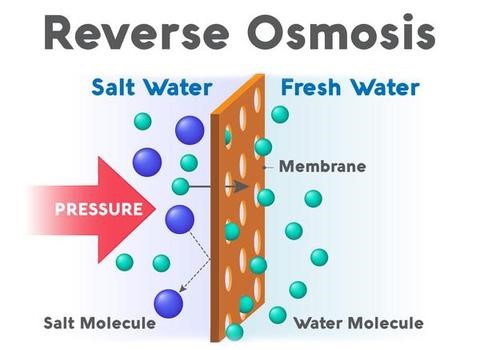
Osmosis is a natural process that balances two bodies of water separated by a semipermeable membrane. Water flows through the semipermeable membrane from the less concentrated solute (fewer contaminants) to the highly concentrated (more contaminants) to restore equilibrium between the two sides. A semipermeable membrane has small holes that trap contaminants but allow water to flow through. This flow of water may be reversed if pressure is applied to the solute with higher concentration.
What is reverse osmosis?
Reverse osmosis occurs when pressure is applied to solute with a high concentration of contaminants. For example, when pressure is applied to a volume of saltwater, the salt is left behind when the water passes to the other side of the membrane. Now, the saltwater is drinkable. The fresh water produced is called the permeate. The concentrate left over is the waste or brine.
Unfiltered water, or feed water, flows to the more concentrated side of the RO membrane, where pressure is applied. The pressure forces the water through the semipermeable membrane. Both the contaminants and the membrane contain ions with like charges. Since like charges repel, the contaminants cannot cross to the other side of the membrane.